Resources
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
With the single photon sensitive EMCCD cameras, Andor have delivered a dedicated, truly high-end, yet accessible scientific camera platform, designed specifically to drive the absolute best from ultra-sensitive EMCCD technology across all critical performance specs and parameters.
NEW iXon Ultra Blue | The World's Most Sensitive Camera for Blue & Near UV Detection.
| Key Specifications | iXon Ultra 888 (inc. Blue) | iXon Ultra 897 (inc. Blue) | OCAM2K | OCAM2S |
| Core Attributes | Field of view, sensitivity and speed | Sensitivity and speed | Ultra-high speed | Ultra-high speed and fast electronic shuttering |
| Sensor format | 1024 x 1024 (Blue 1018 x 1018) |
512 x 512 (Blue 506 x 506) |
240 x 240 | 240 x 240 |
| Pixel Size | 13 µm | 16 µm | 24 µm | 24 µm |
| Sensor Diagonal | 18.8 mm | 11.6 mm | 6.9 mm | 6.9 mm |
| Read Noise | < 1 e- with EM Gain | < 1 e- with EM Gain | < 1 e- with EM Gain | < 1 e- with EM Gain |
| Cooling | -95C | -100C | -45C | -45C |
| Darkcurrent | 0.00011 e-/pixel/sec | 0.00015 e-/pixel/sec | <20 e-/pixel/sec | <20 e-/pixel/sec |
| Spurious Background (CIC) | 0.005 events/pixel | 0.0018 events/pixel | N/A | N/A |
| QE Options | BV, EXF, UVB (Ultra); NBB (Ultra Blue) | BV, EXF, UVB (Ultra); NBB (Ultra Blue) | Back-illuminated deep depletion | Back-illuminated deep depletion |
| Frame Rate | 26 fps (670 fps with 128x 128 Crop Mode) | 56 fps (595 fps with 128x 128 Crop Mode) | 2067 fps full frame | 2068 fps full frame |
| Pixel Well Depth | 65,000 e- | 145,000 e- | 270,000 e- | 80,000 e- |
| Embedded Electronic Shutter | N/A | N/A | N/A | Yes |
| Interface | USB 3.0 (additional Camera Link Output) | USB 3.0 (additional Camera Link Output) | Camera Link® Full | Camera Link® Full |
| Learn More | Specifications | Specifications | Specifications | Specifications |
| Contact Us | Request Pricing | Request Pricing | Request Pricing | Request Pricing |
The single photon sensitive iXon Ultra EMCCD represents the best rapid imaging detector for knowing with a very high degree of certainty whether a photon has been detected in a pixel, especially critical for quantum entanglement experiments. Back-illuminated EMCCD remains the leading technology for determining one from zero.

Both iXon Ultra models offer ‘2 in 1’ performance flexibility, operating either as a single photon EMCCD or a low noise conventional CCD, readily user-selectable through software. Such versatility is attractive in laboratories that can require the camera to operate in low light conditions under both fast and slow frame rate conditions. In photon starved applications, choosing the EMCCD amplifier usually yields better signal to noise ratio when under faster frame rates conditions (> 1 fps), whereas often the CCD amplifier can yield better signal to noise ratio when longer exposures can be applied and when the sensor can be read out slowly (i.e. ‘seconds per frame’ rather than ‘frames per second’).
However, in either EMCCD or CCD configuration, a special ‘Fast Kinetics’ capability can be accessed to collect bursts of data with microsecond time resolution. This can be useful in BEC experiments, for example, to measure time-of-flight rapid cloud dynamics immediately after the MOT is turned off.
| EMCCD | CCD |
| Single Photon Sensitive | 3 to 6 e- read noise |
| Multiplication noise | No Multiplication noise |
| Faster frame rates possible | Restricted to slower frame rates |
The basic trade-offs between EMCCD and conventional CCD amplifiers
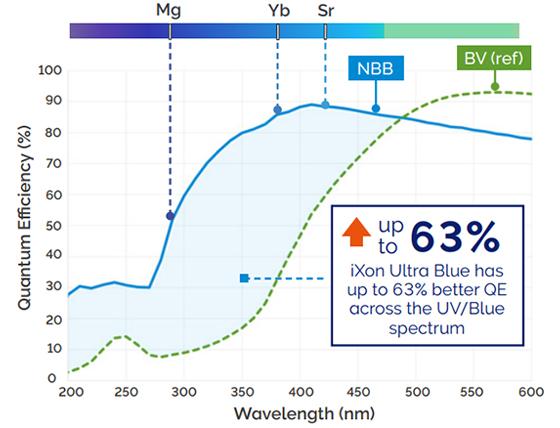
Reaching over 95% peak QE and offering broad wavelength coverage from UV through to NIR, iXon Ultra and iXon Ultra Blue spectral response is suited to a wide range of science; from NIR photometry of cold dwarf stars to single molecule biophysics to UV quantum imaging of trapped ions. Fringe suppression is also available for NIR imaging with reduced etaloning.
The NEW iXon Ultra Blue is the world’s most sensitive detector of blue and near UV photons, combining single photon sensitivity with significantly enhanced QE across the 200 – 450nm range. As such it is ideally suited to imaging of ion trap quantum systems, especially for ions such as Ytterbium (370nm), Magnesium (285nm) or Strontium (422nm).