The very reactive hydroxyl radicals (OH radicals) are the key species in almost all photochemical reactions which are responsible for the degradation and the removal of trace gases from the atmosphere. In the early morning hours the main source of OH radicals is the near-UV photolysis of nitrous acid (HONO)
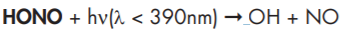 which has been formed in the troposphere during the night. Unfortunately, the gas phase and heterogeneous reactions which produce HONO are only poorly understood. In order to investigate the dominant atmospheric HONO formation and destruction pathways it is essential to measure HONO in the field under different chemical conditions (clean air to mega cities) using sensitive and specific HONO detection techniques. In this project a new optical instrument based on the principle of Incoherent Broad-Band Cavity-Enhanced Absorption Spectroscopy (IBBCEAS) was developed and applied to measure HONO in the Jülich Atmosphere Simulation Chamber SAPHIR.
which has been formed in the troposphere during the night. Unfortunately, the gas phase and heterogeneous reactions which produce HONO are only poorly understood. In order to investigate the dominant atmospheric HONO formation and destruction pathways it is essential to measure HONO in the field under different chemical conditions (clean air to mega cities) using sensitive and specific HONO detection techniques. In this project a new optical instrument based on the principle of Incoherent Broad-Band Cavity-Enhanced Absorption Spectroscopy (IBBCEAS) was developed and applied to measure HONO in the Jülich Atmosphere Simulation Chamber SAPHIR.
Instrumental
In our IBBCEAS instrument, the light from a bright incoherent source (e.g. a short-arc lamp, high power LED, etc.) is transmitted through an optically stable cavity and dispersed with a grating spectrograph and detected by a charged coupled device (CCD) detector. The measurement principle combines the simplicity and robustness of conventional optical absorption spectroscopy with the enhancement concepts developed in Cavity Ring-Down Spectroscopy. The broad simultaneous spectral coverage allows multiple species to be detected and interfering atmospheric constituents to be accounted for.
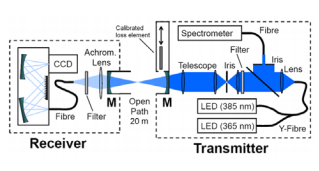
Fig. 1: Instrumental layout of the BBCEAS instrument
A schematic of the open-path setup is shown in the figure (compare Venables et al. (2006), Gherman et al. (2008), and Varma et al. (2009)). The instrument consisted of a transmitter unit and a receiver unit, each containing one of the cavity mirrors M. Both units were placed at either end of the SAPHIR chamber so that the mirror separation was d = (20 ± 0.02) m. The dielectric cavity mirrors (Layertec GmbH) had a reflectivity of about 0.9983 at 375 nm. The transmitter unit housed two temperature stabilized UV LEDs (Omicron GmbH), whose output was combined by means of a branched light guide to cover a spectral range from ~350 to ~400 nm. The emission of the LEDs was optically filtered (Semrock, FF01-377/50-25) before the cavity and monitored during measurements by coupling a fraction of the light into a small reference spectrometer (Avantes AS3648).
In the receiver unit the light was focused into a fiber bundle and connected to the 25 µm entrance slit of a spectrograph (Andor Shamrock SR-303i-A, 1200 lines/ mm grating, spectral resolution 0.21 nm) attached to a cooled CCD detector (Andor Newton DU920P-BU, UV enhanced back illuminated, 1024 x 255 pixels, size 26 x 26 µm2). The mirror reflectivity was regularly calibrated using a calibrated optical loss element in the transmitter unit, which could be moved into and out of the beam path. An acquisition time of 1 min was used for all measurements.
The high sensitivity of IBBCEAS is due to the long effective absorption path length, which is generated by multiple reflections inside the optical cavity. The maximum transmission of the empty cavity depends only on the reflectivity, R, of the mirrors. The effective path length of the instrument equals d/(1-R), where d is the mirror separation. Thus, for mirrors with a reflectivity of R = 0.998 and a separation of d = 20 m, the effective optical path length equals 10 km.

Fig. 2: The Atmosphere Simulation Chamber SAPHIR is a tool for the quantitative experimental investigation of tropospheric chemistry under natural conditions. Design features: double-wall Teflon foil cylinder, 5 m diameter and 20 m length, 270 m3 volume. The optical path of the BBCEAS instruments runs along the central axis of the chamber.
Data evaluation
The extinction of the gas mixture (e) as a function of wavelength (λ) is analysed by means of a linear least square algorithm in order to derive number densities of gas constituents absorbing in the near-UV (HONO and primarily NO2):
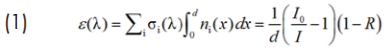
In eq. (1) σi is the wavelength-dependent absorption cross-section [cm2 molecule-1] of the i-th species and ni its number density [molecules/cm3]; I, and I0 are the transmission intensities of the cavity with and without the absorbing target species, respectively, and R is the mirror reflectivity.
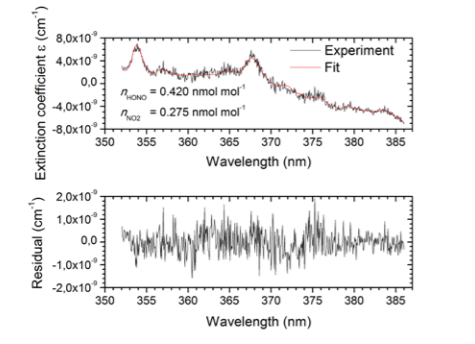 Fig. 3: Upper Panel: Typical absorption spectrum measured with the IBBCEAS system at the SAPHIR chamber including a fit of eq. (2) to the measured data. Lower Panel: Fit residuals.
Fig. 3: Upper Panel: Typical absorption spectrum measured with the IBBCEAS system at the SAPHIR chamber including a fit of eq. (2) to the measured data. Lower Panel: Fit residuals.
Here the number densities of HONO and NO2, referred to as nHONO and nNO2 are retrieved by fitting the measured extinction coefficient to the function εfit:
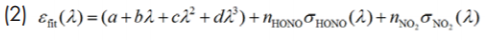 where σHONO and σNO2 stand for the corresponding absorption cross-sections, and a, b, c, and d are the four unknowns of a third-order polynomial accounting for unspecified additional losses (such as background absorption by unknown absorber, Rayleigh and Mie scattering).
where σHONO and σNO2 stand for the corresponding absorption cross-sections, and a, b, c, and d are the four unknowns of a third-order polynomial accounting for unspecified additional losses (such as background absorption by unknown absorber, Rayleigh and Mie scattering).
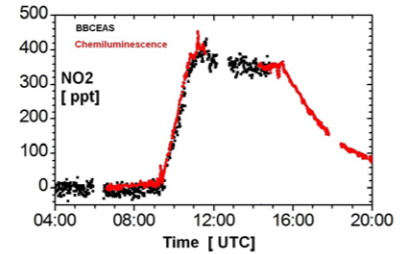
Fig. 4: Comparison of NO2 measurements by BBCEAS and a reference detection technique for NO2 (Chemiluminescence)
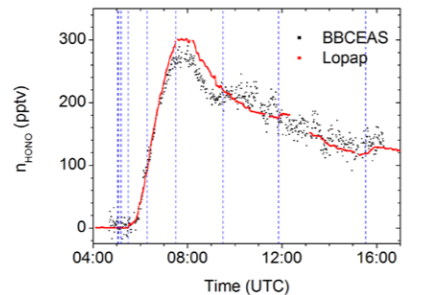
Fig. 5: HONO measurements by BBCEAS and the Long-Path Absorption Photometer (LOPAP) technique.
An overall good agreement for both trend and absolute concentrations was observed between IBBCEAS and the standard instruments available at SAPHIR. The agreement between the time dependent HONO concentrations found with LOPAP and with IBBCEAS was within the error limits of the optical instruments.
References
[1] Venables, D. S., et al. (2006). “High sensitivity in situ monitoring of NO3 in an atmospheric simulation chamber using incoherent broadband cavity-enhanced absorption spectroscopy.” Environmental Science and Technology 40(21): 6758-6763.
[2] Gherman, T., et al. (2008). “Incoherent broadband cavity-enhanced absorption spectroscopy in the near ultraviolet: Application to HONO and NO2.” Environmental Science and Technology 42: 890-895.
[3] Varma, R. M., et al. (2009). “Long optical cavities for open-path monitoring of atmospheric trace gases and aerosol extinction.” Appl. Opt. 48(4): B159-B171.


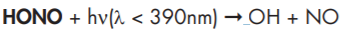 which has been formed in the troposphere during the night. Unfortunately, the gas phase and heterogeneous reactions which produce HONO are only poorly understood. In order to investigate the dominant atmospheric HONO formation and destruction pathways it is essential to measure HONO in the field under different chemical conditions (clean air to mega cities) using sensitive and specific HONO detection techniques. In this project a new optical instrument based on the principle of Incoherent Broad-Band Cavity-Enhanced Absorption Spectroscopy (IBBCEAS) was developed and applied to measure HONO in the Jülich Atmosphere Simulation Chamber SAPHIR.
which has been formed in the troposphere during the night. Unfortunately, the gas phase and heterogeneous reactions which produce HONO are only poorly understood. In order to investigate the dominant atmospheric HONO formation and destruction pathways it is essential to measure HONO in the field under different chemical conditions (clean air to mega cities) using sensitive and specific HONO detection techniques. In this project a new optical instrument based on the principle of Incoherent Broad-Band Cavity-Enhanced Absorption Spectroscopy (IBBCEAS) was developed and applied to measure HONO in the Jülich Atmosphere Simulation Chamber SAPHIR. 

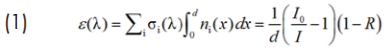
 Fig. 3: Upper Panel: Typical absorption spectrum measured with the IBBCEAS system at the SAPHIR chamber including a fit of eq. (2) to the measured data. Lower Panel: Fit residuals.
Fig. 3: Upper Panel: Typical absorption spectrum measured with the IBBCEAS system at the SAPHIR chamber including a fit of eq. (2) to the measured data. Lower Panel: Fit residuals.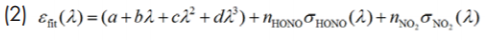 where σHONO and σNO2 stand for the corresponding absorption cross-sections, and a, b, c, and d are the four unknowns of a third-order polynomial accounting for unspecified additional losses (such as background absorption by unknown absorber, Rayleigh and Mie scattering).
where σHONO and σNO2 stand for the corresponding absorption cross-sections, and a, b, c, and d are the four unknowns of a third-order polynomial accounting for unspecified additional losses (such as background absorption by unknown absorber, Rayleigh and Mie scattering). 
