Resources
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
Application Notes
Author: Urban AS, Shen X, Wang Y, Large N, Wang H, Knight MW, Nordlander P, Chen H and Halas NJ Laboratory for Nanophotonics, Rice University
Published: 01 Mar 2014 · Last updated: 16 Jul 2025
Tags: ms-plasmonics-app, microspectroscopy-app
Assembling nanoparticles into well-defined structures is an important way to create and tailor the optical properties of materials. These optical properties of subwavelength metallic structures arise from their ability to support surface plasmons, oscillations of the delocalized electrons in metals that couple with the electromagnetic field.
A universal characteristic of these structures is their exquisite correlation between structural geometry and optical properties, which has provided new approaches to the control and manipulation of light, particularly in the visible and near-infrared regions of the spectrum.[1],[2] This has made it possible to engineer electric and magnetic responses over this wavelength range, enabling the construction of metamaterials with a negative refractive index at these wavelengths,[3],[4] along with a host of artificial media that manipulate light in ways that defeat natural materials.[5],[6],[7]
Most advances in metamaterials research to date have been based on structures fabricated in two-dimensional planar geometries. Although 3-D nanocluster assembly has been reported,[8],[9],[10],[11],[12] cluster stability and robustness has been limited, with disintegration or deformation apparent if removed from the solution or substrate where they were formed or through contact with other solvents or solutions.[9],[13],[14] Aging effects, such as contraction of DNA, may also lead to radical changes in morphology.
In this work, the Rice team shows an efficient method for fabricating highly regular, stable 3-D plasmonic nanoclusters of noble metals by encapsulation within a small polymer sphere that stabilizes their geometry and protects them against a wide range of solvents and solutions. The structural integrity of these clusters allowed an examination of how the optical properties of individual 3-D clusters relate to nanoparticle number, geometry, and orientation of the cluster. Some of the clusters, such as tetrahedra and icosahedra, could serve as the optical kernels for metafluids, imparting metamaterial optical properties into disordered media such as liquids, glasses, or plastics, free from the requirement of nanostructure orientation.
Obtaining small clusters of close packed nanoparticles is quite challenging; the constituent nanoparticles need to be mobile enough to achieve a minimal energy state, yet stable enough to prevent dissociation and aggregation during their isolation and subsequent study.
The Rice team incubated citrate-stabilised 15 nm gold nanoparticles with thiol-terminated polystyrene (PS115-SH) and PS17-b-PAA83 in DMF. The swelling of the long polystyrene chains allows the nanoparticles to maximize their packing within the cluster. Aggregation was induced by adding water to remove the DMF and clusters were directly isolated by centrifugation before characterisation by transmission electron microscopy (TEM) or scanning electron microscopy (SEM).
This fabrication process resulted in a variety of highly regular 3-D clusters containing predominantly 3 - 25 nanoparticles, with an interparticle spacing of nominally 10 nm. In small clusters of three to six particles, the structures formed were triangular, tetrahedral, trigonal bipyramidal, and octahedral, respectively. For larger clusters of 7 to 20 particles, the structures follow an essentially icosahedral growth scheme, with the structures either assembling en route to, or growing onto, an icosahedral cluster. Interestingly, the yield of icosahedral (N = 13) and double-icosahedral clusters (N= 19) is significantly larger than the yield of even slightly differently sized clusters and is directly reminiscent of “magic number” atomic clusters.
Dark-field scattering spectra of specific individual monomers, dimers, trimers, and tetrahedra of the nanoclusters were acquired. ITO-coated float glass substrates were patterned with gold/titanium finder grids comprising 100 x 100 μm squares to repeatedly locate individual nanoparticles in both dark field and scanning electron microscopes. Following dilution to enable imaging of single clusters, 1.5 μL of solution was drop-cast on each sample and allowed to dry. The substrates were illuminated with a quartz halogen aluminium reflector and imaged in an oil-immersion, transmission dark field microscope with a 40x 0.6NA objective (Olympus Corporation). Spectral analysis was provided by a hyperspectral system, comprising a diffraction grating spectrophotometer with automated sample stage (CytoViva technology) with a back illuminated, deep depletion CCD (iKon-M DU934PBR- DD, Andor Technology). The camera cooling was maintained at -80ºC and a total of 251 image slices were acquired at 4 slices per second, with nanoparticle spectra automatically generated through specially written software from Igor Pro (WaveMetrics).
A dark-field scattering spectrum for each individual nanocluster was recorded (Figure 1a, b) and compared with finite element method calculations of the theoretical scattering spectra (Figure 1d). Each individual nanocluster was also imaged by scanning electron microscopy (Figure 1c).
Among these small 3-D clusters, three primary characteristics emerged.
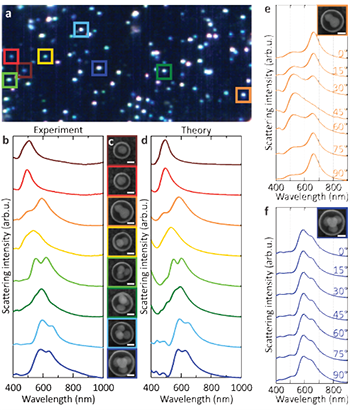
Figure 1: Optical properties of small (N ≤ 4) nanoclusters. (a) Darkfield scattering hyperspectral image of nanoclusters performed over 100 × 100 μm squares, (b) Small (N ≤ 4) silver nanoclusters dark-field scattering spectra. Corresponding SEM images are shown in c. (d) Finite element method calculations of the scattering spectra of the individual nanoclusters (e) Polarization-dependent scattering spectra of the dimers (f) Polarization-dependent scattering spectra of the tetrahedron. Scale bars correspond to 50 nm.
Finite element method (FEM) calculations using the geometric parameters of the SEM images were adjusted in respect of shape, size, substrate and symmetry to account for experimental observations. The observed spectral red-shift of the monomer was shown to be consistent with the presence of a thin silver oxide shell around the nanoparticles. However, the presence of an Ag oxide shell accounted for only 20% of the experimentally observed broadening of the monomer plasmon resonance. An additional contribution to the broadening is the diffusion of silver ions and impurities into the surrounding PS-PAA polymer during the cluster formation process. Indeed, a close agreement with the experimental scattering spectra for the monomer resulted when the PS-PAA polymer was modelled as a lossy medium with n~ = n + iκ, where n = 1.6 and κ is in the range 0.04-0.38. Using this observation as a starting point, the team obtained excellent agreement between the calculated and measured scattering spectra for the clusters shown in Figure 1b-d.
The clusters, while supported on a substrate for the purposes of optical characterisation, retain a random, 3-D, out-of-plane orientation due to their round polymer capsule. This 3-D orientation induces profound changes in the observed optical properties and is shown dramatically in the scattering spectrum of the dimer and trimer clusters, where any change in orientation of the cluster with respect to incident light polarization reveals large spectral shifts and the appearance of additional modes for specific orientations. Extensive angle-dependent calculations, incorporating slight asymmetries due to nanoparticle position and shape within the cluster, were needed to obtain this level of quantitative agreement, even for the simplest cluster geometries. The polarization dependence of the dimer and tetrahedral clusters was examined in greater detail by inserting a linear polariser into the emission path of the microscope and acquiring polarisation-dependent scattering spectra.
The highly orientation-dependent scattering spectrum of the dimer, comprised of linear combinations of transverse and longitudinal plasmon modes, could be clearly observed for this randomly oriented case (Figure 1e). In contrast, the scattering spectrum of the tetrahedron is nearly isotropic (Figure 1f). This observed isotropy provides direct evidence that a plasmonic tetrahedron does indeed behave like an isotropic “metamolecule”: such clusters can be dispersed in a liquid and result in a metafluid.
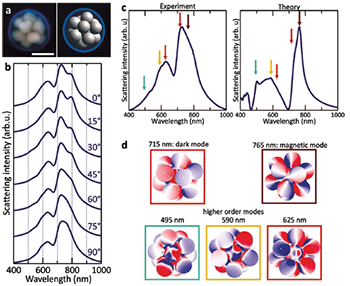
Figure 2: Optical properties of icosahedral clusters. (a) SEM image (left) and 3-D model (right) of an icosahedron (13 NPs). The scale bar corresponds to 100 nm. (b) Polarization-dependent scattering spectra of the icosahedron. (c) Experimental scattering spectra from the FEM calculations. (d) Charge plots of the modes identified in the scattering spectrum of the icosahedron.
As previously mentioned, determining the structure of larger nanoclusters is quite challenging. However, geometrical information can be extracted by comparing their measured optical properties to the calculated optical properties of modelled nanoclusters. To illustrate this possible approach, the Rice team chose a nanocluster which appeared to be an icosahedron in SEM images, another nanocluster with isotropic optical properties (Figure 2a). Indeed, the polarizationdependent scattering spectra of this far more complex cluster vary only weakly with polarization angle (Figure 2b) and are in good agreement with theoretical simulations for a perfect icosahedron (Figure 2c).
Five main localized surface plasmon modes contribute to the calculated spectrum of the icosahedron, as can be seen in the charge plots calculated for each mode (Figure 2d). The two main peaks are associated with a magnetic plasmon mode at 765 nm and a dark plasmon mode at 715 nm. The other three modes, located between 495 and 625 nm, are higher order modes, which can be seen from the calculated charge distributions (Figure 2d). All of these modes show up in the experimental spectrum at approximately the same positions as in the calculations. The observed variations in the relative peak intensities between the experiment and the simulations can be explained by slight structural imperfections in the fabricated structure, mostly arising from variations in shape of the constituent nanoparticles. The team’s conclusion from this direct comparison was that this nanocluster is an icosahedron.
These nanoparticle clusters open up access to a novel breed of optically active materials. The precise way in which the nanoparticles are arranged, rather than their composition, dictates the optical characteristics through an effect called localized surface plasmon resonance. Clusters with highly orientation-independent optical properties, such as tetrahedra and icosahedra, could enable polarization-independent and non-directional negative index media like fluids, free-form solids and isotropic films.
The universality of the fabrication method could extend the use of plasmonic nanoclusters to other regions of the spectrum, either by incorporating different materials, for example, aluminium for the UV; or spherical core/shell nanoparticles, for example, nanoshells for the IR. They are also compatible with easily applied material coating methods, such as aerosols, and could lead to materials with transparency windows at specific frequencies and with constant ratios and linewidths. They could also enable electromagnetic characteristics not yet achievable in current types of metamaterials as well as new approaches to current technological challenges, such as high-throughput chemical and biological sensing.
Metamaterial engineering provides exciting prospects for future photononics applications, such as, the fabrication of ’super-lenses’ that can potentially overcome the diffraction limit associated with traditional lenses.
The structural stability of the nano-bricks at the basis of these metamaterials is critical to ensure consistent and reliable, specific optical properties. For example, knowing that the optical response of an ideal tetrahedron should be virtually independent of cluster orientation is of little use if the 3-D plasmonic nanocluster is unstable. Encapsulating the 3-D nanoclusters in a small polymer sphere stabilizes their geometry, makes them resistant to a wide range of solvents and solutions, and paves the way for the development of novel isotropic metamaterials, such as metafluids.