Resources
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
Application Notes
Author: Valter Kiisk, Michal Novotný, Margus Kodu, et al.
Published: 01 Mar 2025 · Last updated: 01 Apr 2025
Luminescent thin films have a multitude of applications in displays, lighting, photonics and sensing. Metal oxides doped with photostable impurities (such as rare earth ions) constitute a broad class of photoluminescent (PL) materials. Annealing of the as-deposited films is critical to activate the PL centers in a crystalline phase. However, prolonged thermal annealing may cause an undesirable migration of the impurity ions or damage the substrate.
Hereby we demonstrate pulsed laser annealing of rare earth doped titania (TiO2) films [1]. If the laser wavelength lies in the ultraviolet region, the laser light is almost entirely absorbed by the thin film due to a strong band-to-band absorption. As a result, one can achieve an instantaneous localized heating of the film, applicable even on delicate substrates.
Interestingly, the rare earth doped TiO2 nanomaterials pose a novel chemical sensing capability. It has been proposed that the adsorption/desorption of oxygen species affects the excitation or emission of the dopant ions through a charge transfer process [2,3]. Such optical gas sensing has potential advantages over the conductometric principle, being less affected by electromagnetic interference and enabling non-contact operation.
A pulsed excimer laser (Coherent COMPex Pro 205, wavelength 193 or 248 nm, pulse duration 20 ns) was used both for thin film deposition as well as annealing of the as-deposited film. Samarium (Sm) and neodymium (Nd) doped TiO2 films on SiO2 or SiO2/Si substrates (10×10 mm2) were deposited at room temperature by ablating ceramic TiO2:Sm and TiO2:Nd targets in an oxygen atmosphere (Fig. 1a). About 40000 laser pulses resulted in a film thickness of ~150 nm. In the annealing setup, the laser beam was incident on the sample at 70° angle (Fig. 1b). Laser fluences 23 and 50 mJ/cm2 were tested, with 1–100 laser shots applied to the sample. Optical properties were monitored in-situ, using fiber-coupled instruments [4]. For in-situ PL, the sample was excited with the same laser at a reduced fluence.
Notable PL intensity emerged only when sample was treated at 50 mJ/cm2 (Fig. 1c). At this fluence, ~10 laser pulses were sufficient to optimize PL intensity. Excessive number of laser pulses started to reduce PL intensity and initiated anatase-to-rutile phase transition (as revealed later by Raman spectroscopy).
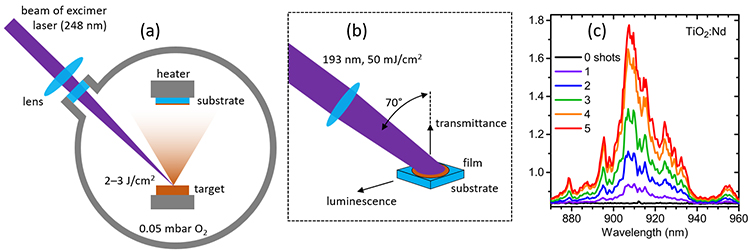
Figure 1. (a) Pulsed laser deposition of thin TiO2 film. (b) Laser annealing with in-situ monitoring of transmittance and luminescence. (c) Evolution of in-situ measured PL spectrum of TiO2:Nd film with increasing number of laser shots at 50 mJ/cm2.
While the deposited film covered a circular area of 10 mm in diameter, the annealing laser beam could not be made uniform across such a wide area. The lateral homogeneity of annealing was best evaluated via PL imaging. A low-intensity pulsed beam from the excimer laser was expanded to yield an even illumination of the whole sample whereas time-gated (delay 5 µs, width 1000 µs) PL images were acquired by cooled Andor iStar 18U-03 ICCD camera through a Helios 44M objective lens (Fig. 2a). While the overall gradation of the sample is visible already in reflection/transmission, the majority of the film area still yields a reasonably uniform PL intensity (Fig. 2b).

Figure 2. (a) Setup for PL imaging of the sample. The PL was excited by an expanded 248 nm pulsed laser beam, uniformly illuminating the sample, whereas an intensified CCD camera captured the time-gated image. (b) Optical images of the laser annealed TiO2:Sm sample (50 mJ/cm2, 20 shots) under ambient light (top) and in luminescence (bottom).
Raman and luminescence microspectroscopy studies showed that, compared to a sample thermally annealed at 700 °C, the optimally laser treated areas exhibited an order of magnitude improvement in terms of host-sensitized PL intensity and crystallization of the film (nearly phase-pure anatase) [1].
For gas sensitive PL measurements, the TiO2:Sm3+ sample was placed on a heater block inside an airtight chamber with gas feedthrough and a quartz window (Linkam THMS350V). Mass-flow controllers were used to establish a constant flow of oxygen/nitrogen mixture through the chamber. The central area of the sample was illuminated by 365 nm light emitting diode (Thorlabs M365LP1), which provided continuous and stable excitation at an estimated irradiance ~16 mW/cm2. The LED was spectrally cleaned with appropriate band-pass filters. The emitted PL was collected in a backscattering geometry, dispersed by a monochromator (LOMO MDR-23) and recorded by Andor iDus DU420-BU CCD camera, which was operated in full vertical binning mode and a 1 s exposure time. Data acquisition was organized by a script running in the Andor SOLIS environment. The script acquired a background-corrected spectrum every second, integrated the areas under the main Sm3+ emission bands, and saved the data into a file.
While monitoring the PL intensity, the sample was repeatedly exposed to a series of small concentrations of oxygen (Fig. 3). During each cycle, oxygen concentration increased stepwise in sequence 0.02-0.05-0.1-0.2-0.5-1.0-2.0 %O2 (10 min each) and then switched back to 0% O2. In terms of sensitivity and power consumption, such optical sensor yields an excellent response: it can detect 100 ppm O2 concentrations while operating at room temperature. Preliminary studies show that the material responds to even smaller concentrations of polluting gases, such as NH3.
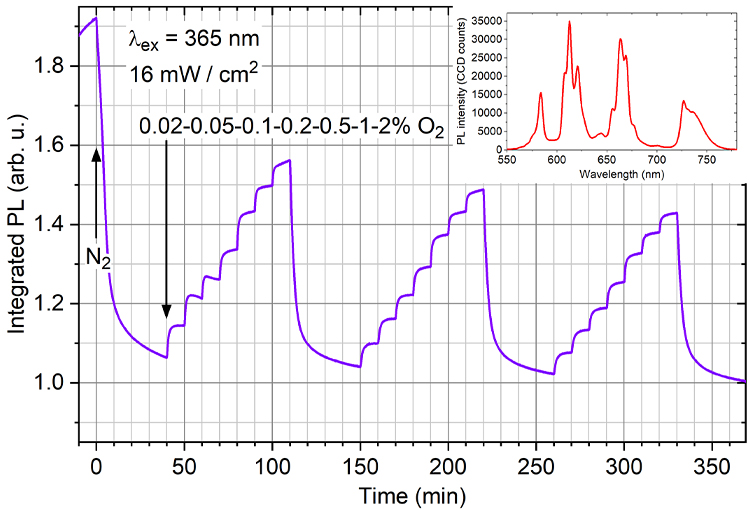
Figure 3. Optical trace oxygen sensing with laser annealed TiO2:Sm3+ films at room temperature. Once every second, the PL spectrum of Sm3+ (shown in the inset) was recorded and the areas under the emission bands integrated. The sample was initially exposed to multiple cycles of pure oxygen."
There remains an issue with stability, as evidenced by the gradual drift of the PL signal over the timescale of several hours. By subjecting the sample to special conditions prior to the main gas sensing cycle, it was found that the baseline drift relates to some very slow adsorption-desorption of oxygen [1]. Further improvement of sample preparation is needed to reduce the heterogeneity of the sensor surface.
[1] V. Kiisk, et al., Applied Surface Science 637, 157884. (2023), DOI: https://doi.org/10.1016/j.apsusc.2023.157884.
[2] M. Eltermann, et al., Sensors and Actuators B: Chemical 265, 556 (2018), DOI: https://doi.org/10.1016/j.snb.2018.03.034.
[3] V. Kiisk, et al., J. Phys. Chem. C 123, 17908 (2019), DOI: https://doi.org/10.1021/acs.jpcc.9b04588.
[4] M. Novotný et al., Materials 14, 7576 (2021), DOI: https://doi.org/10.3390/ma14247576.