Resources
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
Case Studies
Author: Dr Claudia Florindo and Inês Baião-Santos
Published: 01 Mar 2022 · Last updated: 11 Sep 2025
Cell division is essential to life, and errors in this process can lead to cell death or genomic instability which is associated with cancer development. However, how cell proliferation is regulated is still not fully understood.
In brief, somatic cell division (mitosis) is the process by which one cell divides to give rise to two genetically identical daughter cells. Key players in this process are the centrosomes, microtubules, and chromosomes. These structures join in an orchestrated and tightly regulated manner to quickly accomplish faithful chromosome segregation. Successful mitosis is achieved by the ordered interplay of kinases, phosphatases and proteolysis.
Polo is an essential mitotic kinase first discovered in Drosophila melanogaster (Llamazares et al., 1991) and later found to be conserved across all the eukaryotic kingdom. The first polo homolog to be described in mammalian cells was the Polo-like kinase one (PLK1) which is a central regulator of cell division. Plk1 kinase acts in several critical points of mitosis, being some of its functions:
Movie 1 – Live imaging of cell division (Mitosis) observed with Andor Benchtop Confocal - BC43. Mammalian cells were imaged with BC43 using confocal imaging mode for over 6 hours. At each time point, 8 independent positions were imaged, and for each position, we acquired 3 channels and 15 Z stacks. The movie shows one of the 8 positions. It can be observed the cell going in and out of mitosis by analysing the microtubules (Blue) and the DNA (Yellow. (Yellow – DNA, Blue - Microtubules, Red - Sir Actin). Image credits: Ines Baião-Santos, Álvaro Tavares – Universidade do Algarve, Claudia Florindo – Andor Technology
Another relevant protein family in the control of cell proliferation is the MOB1-like protein family. MOB1-like proteins (MOBs) are kinase activators and, as the PLK kinases, are conserved throughout the eukaryotic kingdom. Both PLK1 and MOB1 have a dynamic localisation through the cell cycle. Both localise at the centrosome at the beginning of mitosis and at the midbody just before cytokinesis. MOB1 localisation is characteristic of key cytokinetic regulators. In fact, both PLK1 and MOB1 are essential for the completion of cytokinesis.
Movie 2 – Live imaging of early-stage divisions in Drosophila embryo with Andor benchtop confocal. Drosophila syncytial embryos were imaged using the confocal imaging modality of BC43, the video is a MIP of 3 Z-stacks showing in cyan GFP-polo and in Yellow Histone 2B-mcherry. The use of GFP-polo as a marker allows the dynamic visualisation of mitosis showing the centrosomes (at early stage of division) and both the centrosomes and spindles (from prometaphase till anaphase). The GFP-Histone 2B allows to phenotypically address the DNA in mitosis and access (among other features) DNA miss segregation. (in Cyan -GFP polo, in Yellow-Histone 2B-RFP). Image credits: Ines Baião-Santos, Álvaro Tavares – Universidade do Algarve.
MOB1 acts in two cellular pathways, the Mitotic Exit Network (MEN) in yeast, and in the Hippo pathway in metazoans. The MEN controls the timely execution of mitotic exit. The Hippo pathway controls the coordination between the cell cycle and tissue growth (organ size control and regeneration). Notably, all the core components of the Hippo signalling cascade were shown to be tumour suppressor genes, and MOB1 is no exception. The de-regulation or function impairment of Hippo pathway components and, in particular, of MOB1, leads to tumorigenesis.
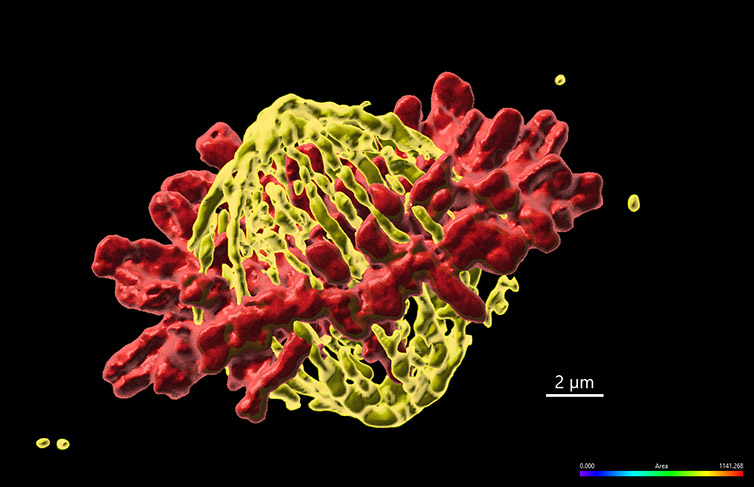
Figure 1 – Mammalian cell in metaphase imaged with Andor benchtop confocal - BC43. Widefield Imaging modality was used to image fixed mammalian cells. The image shows 1 of the 20 independent positions acquired in this experiment. We have acquired two independent channels for each position and covered a range of 15 µm. The deconvolution option was activated in the protocol. Images were further processed in imaris, and we present the MIP of a surface rendered image. (Yellow - Microtubules, Red-DNA). Image credits Ines Baião-Santos, Álvaro Tavares – Universidade do Algarve, Claudia Florindo – Andor Technology.
Cell biology studies that identify and scrutinise the unknown molecular function of genes foster new knowledge. The acquired knowledge can (and is often) used in applied science to improve human health (e.g., by promoting the development of new therapies).
Significantly, several key cell cycle regulators are currently undergoing active clinical trials addressing cancer therapies (among other diseases). The PLK1 gene is one such example. The NIH clinical trial database currently shows 27 studies analysing the efficacy of PLK1 inhibitors as cancer therapeutic agents or PLK1 as a cancer prognostic marker.
In summary, the complete regulation of the cell cycle is not yet understood, neither the complete molecular function of the tumour suppressor MOB1 and the oncogene PLK1. Therefore, a better understanding of the cell cycle regulatory mechanisms will undoubtedly reveal more insights for future cancer therapies.
Álvaro Tavares is the group leader of the Cell Cycle and Cancer Biology laboratory at Universidade do Algarve. The underlying theme behind the Tavares laboratory is the basic biology of mitotic cell division, particularly the aspects regulating the formation of a bipolar mitotic spindle and the connection between centrosomes and cytokinesis.
Another focus area of the lab is the research on the function of proteins that drive other non-directly mitotic cellular functions, for example, cell death, cell migration or uncontrolled cell proliferation. The goal of the lab is to understand how modifications of these cellular processes contribute to the conversion of normal cells into cancer cells.

Figure 2 – Tavares lab photo. From left to right Daniela Silvestre, Inês Lousa, Ines Baião-Santos, Juan Maraver and Álvaro Tavares (Group Leader).
To achieve its research goals, the lab takes advantage of Drosophila melanogaster and human cultured cells using a combination of biochemical, genetic and cytologic techniques. Examples of some of the technologies used in the lab are:
Professor Tavares used the new Andor Benchtop confocal imager - BC43 in his research, and we asked him about his experience when imaging cell division with the system:
"It is very compact and effortless to use. I found that the Andor Benchtop delivers high-quality imaging for filming live cells. The system is capable of producing extraordinary results either in fixed or live samples."
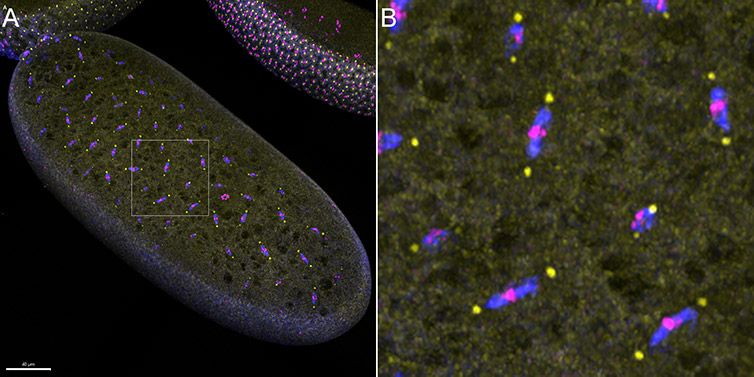
Figure 3 - Confocal Imaging modality of Andor benchtop confocal was used to image early-stage divisions of the Drosophila embryo. A) The image presented is a result of the stitching of 6 imaging fields, over a Z range of 50 µm. B) Magnification of the area highlighted within the white square in A) shows the details of the early Drosophila embryo mitotic spindle. (Blue-Microtubules, Yellow-centrosomes, Magenta-DNA). Image credits: Ines Baião-Santos, Álvaro Tavares – Universidade do Algarve, Claudia Florindo – Andor Technology.
Cell division accuracy is critical to maintain cell ploidy and genomic stability. It relies in the correct establishment of a cell division axis, which is under the control of the cell´s environment and its intrinsic polarity. The Tavares laboratory addresses how cell proliferation is regulated in high eukaryotic cells following two independent lines of research: 1) Mitotic spindle assembly and function and 2) Analysis of the role of Mob-like proteins as central players in cell proliferation.
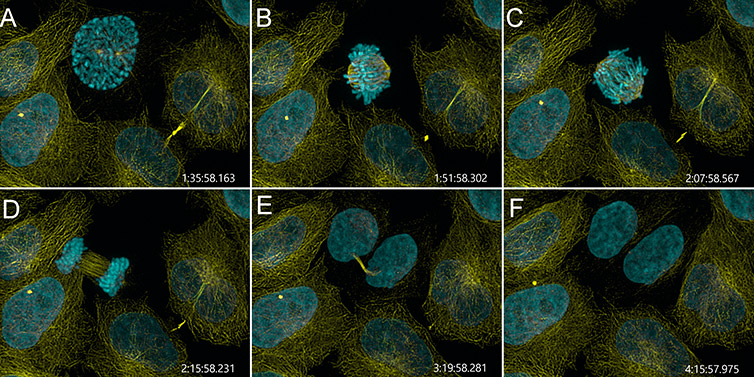
Figure 4 – Mammalian cell division observed with Andor Benchtop Confocal. Mammalian cells were imaged with BC43 using confocal imaging mode for over 4 h. At each time point, 4 independent positions were imaged and for each position 3 channels and 15 Z stacks acquired. We present stills of a movie from one of the 4 independent positions showing one cell advancing through the four mitotic stages: prophase (pannel A), metaphase (panel B), anaphase (pannel C) and telophase (pannel D). (Yellow-microtubules, Cyan – DNA, Time in h:min:sec:ms). Image credits: Ines Baião-Santos and Álvaro Tavares, Universidade do algarve, Claudia Florindo, Andor Technology.
The centrosomes establish the mitotic spindle poles and defects in centrosome number or structural organization can lead to abnormal chromosome segregation. The highly conserved Ser/Thr kinase Polo-like kinase 1 (PLK1) has essential roles in the assembly of a functional bipolar spindle with focused poles.
Professor Tavares have first found that PLK1 (polo) is required for centrosome maturation and cytokinesis (Llamazares et al., 1991). This kinase turned out to be a critical regulator of the cell cycle progression and generated a great interest in the scientific community. Although it has been the object of intense research, the mechanism by which PLK1 regulates spindle-pole formation is still being detailed. Furthermore, PLK1 kinase was found to be an oncogene and is now under research in several clinical trials as a promising target for cancer treatment or prognostic marker.
The Cell Cycle and Cancer Biology laboratory aims to gain a deeper understanding of key cell cycle regulators such as PLK1. The final goal is to contribute to the development of new and improved anti-cancer therapies.
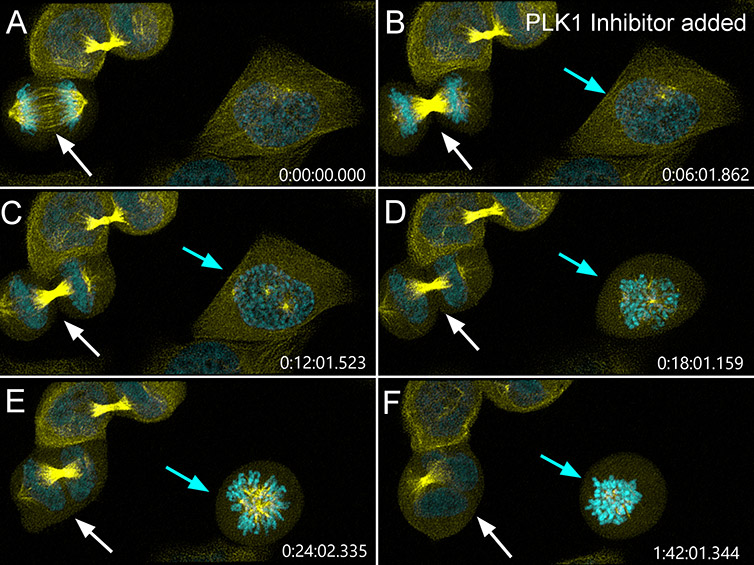
Figure 5 – PLK1 inhibition impairs different stages of mammalian cell division. Mammalian cells were imaged with BC43 using confocal imaging mode using the same imaging conditions as in figure 4. Six minutes after imaging has started (panel B) PLK1 inhibitor (BI-2536, Selleck) was added to the cells. The white arrow shows a cell that was completing cell division when PLK1 inhibitor is added. PLK1 inhibition after anaphase onset (panel B) induces the regression of the cleavage furrow and causes abscission failure (panel D) giving rise to tetraploid binucleated cells (panel E-F) with supernumerary centrosomes (a common feature in tumour development). Highlighting PLK1’s role after anaphase onset (as a regulator of cytokinesis). The cyan arrow shows a cell that is initiating mitosis (panels A-B) when PLK1 inhibitor is added. The two centrosomes (seen in panel C) fail to establish a bipolar spindle upon nuclear envelope breakdown (panel D) and collapse to form a monopolar spindle (panel E). Without proper bipolar attachment, the cell stays arrested in mitosis (panel F) and could go through mitosis or become polyploid by mitotic slippage. (Highlighting PLK1’s role in centrosome function and bipolar spindle establishment and maintenance.) (Yellow-microtubules, Cyan – DNA, Time in h:min:sec:ms). Image credits: Ines Baião-Santos and Álvaro Tavares, Universidade do algarve, Claudia Florindo, Andor Technology.
An example of the importance of analysing the function of critical regulators of the mitotic spindle is the work the laboratory did on the spindly gene. Spindly is required for proper chromosome attachment to spindle microtubules and for chromosome segregation. Paclitaxel is a drug that stabilizes microtubules, and, by doing so, impairs cell division. It is widely used in cancer treatment. Nevertheless, resistance to treatment as well as its high toxicity and secondary effects are important concerns with the use of this chemical.
In collaboration with Dr Hassan Bousbaa (CESPU, Portugal) a combined approach using both a spindly inhibitor and paclitaxel as anti-cancer agents were tested. The results were encouraging and showed that a combined action of spindly inhibitor and microtubule stabilization drug exacerbates cell death response of cancer cells treated with low doses of paclitaxel (Silva et al., 2017).
A second focus area of Professor Tavares Laboratory is the function of MOB-like proteins in cell proliferation. MOB-like proteins are kinase-partners that play roles at different stages of the cell cycle. For example, they are critical components of the Hippo signalling pathway, a pathway that coordinates cell proliferation and organ growth.
The team has proven that MOB1 is required for the final stages of cell division, cytokinesis, by promoting microtubule destabilization (Florindo et al., 2012).
In collaboration with Dr Helena Soares (IGC, Portugal) the centriolar function of MOB1 was further studied using the ciliated protozoan Tetrahymena termophila. The exciting results showed that MOB1 is an intrinsic cell polarity marker, the first molecular polarity marker described in this organism (Tavares et al., 2012). Just like in the mammalian cells, downregulation of MOB1 in Tetrahymena causes a failure to execute cytokinesis. Taken together with the results in mammalian cells, where that lack of MOB1 increases cell motility after cytokinesis and induces prolonged centriole separation in the G1 phase, the group advanced the hypothesis that MOB1 is required for cells to regain full interphasic polarity after division. In this regard, it is noteworthy that downregulation of MOB1 results in tumour formation both in Drosophila and in mice.
Movie 3 - Analysing Microtubule dynamics by tracking the growing tips of microtubules with EB1-GFP. Hela cells expressing EB1-GFP were imaged with BC43 in confocal mode, for 6 min at a frame rate of 2 frames per second. It can clearly be observed the microtubules plus end tips growing from the centrosomes of the cell that is going into mitosis.(Note that: 1- Failures in mitosis can be caused by impairment in microtubule dynamics, 2- Some cancer treatments directly target microtubule dynamics, e.g. paclitaxel). Image credits Ines Baião-Santos, Álvaro Tavares – Universidade do Algarve, Claudia Florindo – Andor Technology.
Importantly, MOB-like proteins are highly conserved in the eukaryotic kingdom. An example of such is the 100% protein identity from mouse to human, suggesting an essential and conserved function. In evolution, its often-found gene duplication events from less complex to more complex organisms. The MOBs were no exception, and in mammals, seven MOB-like genes are present.
Several questions remain to be answered:
The Tavares lab is trying to answer these questions that answer fundamental aspects of cell division. The answer to these questions will undoubtedly open the door to new developments in cancer therapies.
As a final note, Professor Tavares told us that the execution of these studies is heavily dependent on microscopy techniques. We have asked him for a final comment about BC43 performance, and the reply was:
”The Andor Benchtop confocal excelled in the capability to obtain high-quality results in live-imaging of dividing cells. I found it to be a little giant: small in size, giant in image quality. “
| Glossary | |
| Mitosis | Mitosis is the process by which one cell gives rise to 2 daughter cells with equal genetic content. Mitosis is composed of 4 stages (prophase, metaphase, anaphase, telophase) that can be distinguished visually by analysing the microtubules and/or chromosomes (DNA). |
| Paclitaxel | Paclitaxel is a chemical compound that stabilizes microtubules and causes cell cycle arrest. It is used in chemotherapy as a treatment for several types of cancer. |
| Spindly | Spindly is a protein required for proper chromosome segregation by controlling the chromosome bipolar attachment to the spindle. |
| MOB-like genes | MOB like genes is a conserved family of genes from yeast to humans. MOB-like proteins are kinase partners. There are 7 MOB-like genes in Humans. One of such is the tumour suppressor MOB1. |
| Mitotic Spindle | The spindle or mitotic spindle is a macromolecular complex that drives chromosome segregation to the daughter cells. It comprises hundreds of proteins and macromolecular complexes; the microtubules are an essential spindle constituent. |
| Centrosome | The centrosome is an organelle that nucleates the spindle's microtubules and drives the poles' focusing. A dividing cell has two centrosomes, one at each pole of the mitotic spindle. Abnormal centrosome numbers often drive aberrant mitosis and are found in many tumours. |
| Microtubule | Microtubules are polymers of alpha- and beta-tubulin and are a part of the cell cytoskeleton, providing shape and structure. Microtubules are essential constituents of cilia and flagella and are involved in numerous cellular processes such as cell movement, vesicle trafficking, etc. In addition, microtubules are crucial in mitosis; they are an essential component of the mitotic spindle. |
| Kinase | Kinase are enzymes that transfer a phosphate group from a high energy molecule to its substrate. Kinases are involved in regulating complex cellular processes through the phosphorylation of its substrates. Mitosis is highly regulated by kinases. |
| PLK1 (Polo) | PLK1 – Polo Like Kinase 1 PLK1/Polo (polo in Drosophila) is an essential mitotic regulatory kinase whose functions regulates several key points of the cell cycle such as 1) G2/M transition, 2) centrosome maturation, 3) formation of the bipolar spindle, 4) promotion of bi-oriented chromosome attachment and 5) cytokinesis. PLK1 is a proto-oncogene and is overexpressed in several tumours. |
| tumour suppressor gene | Genes in which loss-of-function leads to cellular over proliferation (i.e. tumour development). Tumour suppressor genes typically function as “brakes” to restrain inappropriate cell division and/or to stimulate cell death. |
| Oncogene | Oncogene is a gene that upon acquiring gain of function (by mutation or overexpression) will promote cancer formation. |
| Cytokinesis | Cytokinesis is the final stage of cell division in which the cytoplasm of the cell divides, giving rise to two daughter cells. For proper genome stability, cytokinesis must occur after correct chromosome segregation. |
| Midbody | The midbody is a structure formed at the end of cytokinesis, just before the final stage of cell division – the abscission. The midbody is composed of bundles of microtubules that need to be depolymerized for the final cell separation stage. |
| Hippo Pathway | Evolutionary conserved signalling pathway that controls organ size by regulating cell proliferation and apoptosis. Activation of the Hippo pathway leads to a stop in cell proliferation while deregulation of the Hippo pathway leads to over-proliferation and cancer development. The profiling of human tumour samples shows a widespread deregulation of multiple Hippo pathway components. |
| MEN (Mitotic Exit Network) | The Mitotic Exit Network (MEN) is a signalling cascade originally described in yeast S. cerevisiae, that controls the exit of mitosis and promotes faithful division of cells. |
Bibliography