Resources
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
‘SRRF-Stream+’ is a real time super-resolution microscopy functionality that super-charges the imaging performance of your standard microscope. Attain a level of resolution that was previously only possible from a dedicated high-end Super Resolution Microscope. Based on the “SRRF” technique (Super Resolution Radial Fluctuations), SRRF-Stream+ offers an easy and flexible software approach to super resolution.
NEW – now available for ZL41 Cell, Sona and selected Zyla 4.2 PLUS cameras making this amazing technology accessible to more applications at an even lower price point!
Desmin labelled in cardiomyocytes imaged with a Zeiss Widefield microscope at 60x and Sona 4.2B-6 camera with and without SRRF-Stream+ applied. Images courtesy of Matt Caporizzo, Prosser Lab, Philadelphia, USA.

Image comparison of a fluorescently labelled BPAE cell, recorded with a widefield fluorescence microscope and a SRRF-Stream enabled iXon Life 888 EMCCD camera. A x63 objective was used, with further 2x magnification and 560nm illumination. 100 raw ‘input’ images were recorded for every resultant super-resolution image, resulting in a super-resolution image rate of 0.5 Hz. For a fair comparison without SRRF-Stream, 100 standard widefield images were recorded and then averaged. While the original image was of a larger field of cells, a zoomed ROI of one cell is shown here in order to more easily display a line intensity profile comparison through a small region. The improvement in resolving power is readily apparent.
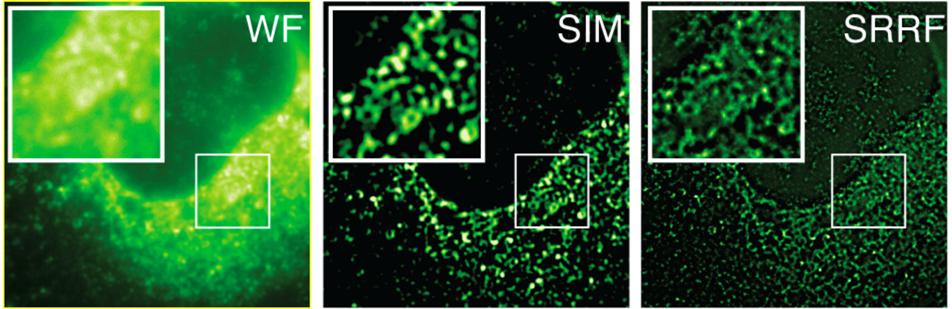
HCV infected cells stained with anti-NS5A. Here we are comparing Widefield (WF), Structured Illumination Microscopy (SIM) and SRRF images (SRRF of the widefield image). The images are of the same field of cells, recorded on the same microscope, using identical objective and optical path. The only difference being that SIM was recorded using an sCMOS detector with 6.5µm pixels whereas the Widefield and resultant SRRF was recorded using an iXon EMCCD detector with 16 µm pixels. The superior resolving power of SRRF is evident, indicative that SRRF is achieving a greater than 2-fold improvement over the classical diffraction limit. SIM is theoretically limited to a 2-fold reduction of the classical diffraction limit. Sample courtesy of the Grove lab at UCL.
F-actin of BPAE cells labelled with Alexa Fluor 488 Phalloidin imaged on a Nikon Ti2 microscope at 60x and Sona 4.2B-11. The image taken from the average of 100 frames compared against the SRRF-Stream+ image. Images courtesy of Motosuke Tsutsumi, Research Institute for Electronic Science, Hokkaido University and National Institutes for Physiological Sciences, Aichi, Japan.
200s time-lapse of a live BSC-40 cell labelled with cell mask and imaged with 635nm LED illumination. The first 100 frames correspond to widefield imaging with a 1 second exposure; the second 100 frames correspond to SRRF-Stream imaging where each frame is produced from SRRF-Stream processing of 50 images (20ms exposure time). Sample preparation by David Albrecht (Ricardo Henriques and Jason Mercer labs at UCL).

Comparative images of Clathrin coated pits of live HeLa cells, labelled with mCherry, recorded on a widefield microscope at 2 FPS. 100 raw ‘input’ images were recorded for every resultant super-resolution image, resulting in a super-resolution image rate of 2 FPS. A line intensity profile is shown through a small region of the SRRF-Stream image, indicating resolution of structures that are 150nm apart. Sample preparation by Caron Jacobs (Ricardo Henriques and Mark Marsh labs at UCL).
Comparative 3D projection montages of fission yeast lifeAct expressing strain. Recorded with standard widefield versus SRRF-Stream widefield, using identical exposure times. Strain courtesy of Mohan Balasubramanian’s laboratory (U. Warwick) Sample courtesy of Gautam Dey (Buzz Baum laboratory at UCL).

Comparative standard widefield and widefield SRRF-Stream images of blood platelets, red membrane, green internal granules. Sample courtesy of Cutler laboratory at UCL.
A still widefield image of a live HeLa cell expressing tubulin-GFP followed by a SRRF-Stream time-lapse of the same region at 1fps (SRRF-Stream analysis of 50 frames at 20ms exposure). Sample preparation by David Albrecht (Ricardo Henriques and Jason Mercer labs at UCL).
With its ability to smash through the classical diffraction limit, and furthermore, to accomplish this in real time, with standard sample labelling, conventional equipment and with low intensity illumination, SRRF-Stream paves the way to unlock previously unseen cellular structure and behaviour, at unprecedented cell-friendly manner.
Adopting the recently developed SRRF technology from the lab of Dr Ricardo Henriques, University College London (UCL), and working in close collaboration with Dr Henriques, Andor have enhanced the technology for optimal performance on SRRF-Stream enabled cameras. Andor are also expert in advanced GPU processing optimization techniques, employed in this instance to execute the SRRF algorithm up to 30x faster than the existing ImageJ-based post processing implementation of SRRF (NanoJ-SRRF). This significant acceleration enables workflow enhancement, by allowing data acquisition and SRRF processing to operate in parallel.
Processing Speed Comparison - SRRF Stream vs NanoJ-SRRF
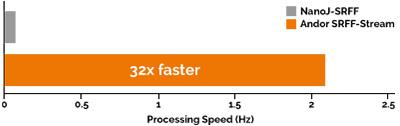
This graph compares the rate of processing of blocks of 100 raw input images (1024 x 1024 pixels), to yield resultant SRRF super-resolution images of 4096 x 4096 pixels. SRRF-Stream is compared to NanoJ-SRRF, the processing occurring on the same Nvidia GTX 1070 GPU card. The SRRF-Stream acceleration subsequently allows data acquisition and processing to happen in parallel, yielding a further workflow improvement over NanoJ-SRRF.
Since processing is now much faster than the camera can acquire data, ‘SRRF-Stream enabled’ cameras now accomplish real time super-resolution, with large field of view super-resolution images.
Having thoroughly tested SRRF-Stream in our own lab, we are very impressed by both the workflow and also the ability to now utilise larger fields of view for live cell super-resolution. By seamlessly combining the SRRF algorithm with the high-performance of the iXon, we have accomplished the world’s first super-resolution camera for fluorescence microscopy.


Yes! If you have a compatible Andor camera: iXon (Ultra and Life EMCCD), Sona, ZL41 cell, Zyla 4.2 PLUS series sCMOS cameras you can unlock SRRF-Stream+ super-resolution microscopy capability.
PLEASE NOTE:

A SRRF-Stream+ compatible Andor camera:
OPTIONAL: PC Workstation - Pre-installed with a recommended and tested GPU card, alongside SRRF-Stream+ enabled MicroManager (linked to the serial number of your Andor camera) and Andor SDK with SRRF-Stream
* Contact your Andor representative with your camera serial number to confirm camera compatibility.
** The Nvidia GPU card should have Compute Capability v3.5 or above and 4GB or greater on-board GPU RAM for iXon, 8GB for Sona, ZL41 Cell and Zyla 4.2 PLUS. Note that Andor have tested ‘mid-range’ cards such as the GTX 1070 and found that this level of card can process data much faster than the rate of camera data acquisition.
Upgrade to SRRF Stream+In order to make SRRF-Stream+ as widely accessible as possible, it has been fully integrated into MicroManager (64-bit) open source microscopy software platform.
The Learning Centre hosts a wide range of technical articles and webinars on various applications and microscopy techniques. Here are some key articles that are related to SRRF-Stream for further reading and how to get the best performance out of this exciting technology!