Resources
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group

Featured Lab - Dr Aparna Lakkaraju
The goal of the Lakkaraju laboratory is to use cell biology to discover novel therapies for blinding diseases such as age-related macular degeneration (AMD). This devastating disease robs people of central high-resolution vision and affects over 30 million older adults worldwide. There are currently no effective treatments for the chronic form of AMD called geographic atrophy, which leads to a slow decline in vision in 90% of patients.
Dr Lakkaraju and her team study the retinal pigment epithelium (RPE), a monolayer of post-mitotic epithelial cells that forms the outermost layer of the retina and performs many functions that are indispensable for vision. A key feature of AMD is the accumulation over time of cellular debris or garbage within and around the RPE, which can compromise RPE function and induce inflammation to promote disease.
The lab focuses on elucidating the retinal pigment epithelium (RPE), the outermost layer of the retina. Each day, to counter the effects of light damage, photoreceptors shed outer segment tips, which are phagocytosed and degraded by the RPE. The substantial number of cells that need to be consumed, coupled with the post-mitotic nature of the RPE, places an enormous burden on mechanisms involved in the removal of cellular garbage in the RPE. With age, incomplete digestion of outer segments leads to the accumulation of vitamin A metabolites called lipofuscin within RPE lysosomes and lipid-protein deposits called drusen above and beneath the RPE. Over a lifetime, these aggregates conspire with environmental and genetic factors to damage the RPE, leading to photoreceptor dysfunction and a slow decline in vision as seen in AMD.
The team utilise multiple tools such as high-speed, high-resolution live imaging of healthy and diseased RPE, biochemistry, genetics and cell and molecular biology to follow critical functions in retinal pigment epithelial cells such as cellular clearance (lysosome function, autophagy, secretion of exosomes), organelle biogenesis and intracellular trafficking.
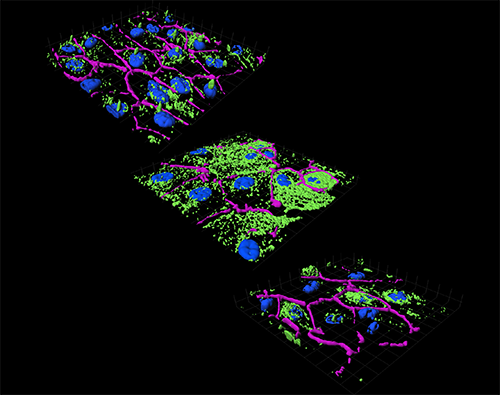
3D reconstructions of acetylated tubulin immunostaining (green) in polarized monolayers of adult primary retinal pigment epithelium (RPE). In normal cells (top panel), acetylated tubulin is mostly localized to the primary cilia. In cells with the vitamin A metabolite A2E, elevated levels of cholesterol and ceramide promote hyperacetylation of tubulin (middle panel). Disruption of microtubule dynamics interferes with autophagosome trafficking and autophagic flux in the RPE. Specific inhibition of acid sphingomyelinase decreases ceramide and acetylated tubulin (bottom panel) and restores autophagy in RPE with A2E. Tight junctions between cells are demarcated by ZO-1 (purple) and DAPI-stained nuclei are blue.
Their current work is focused on answering the following questions:
To address these questions, they use high-speed, high-resolution imaging of live polarized primary RPE cells using Andors Revolution XD spinning disk/TIRF microscopy, coupled with powerful 4D image analysis using the Imaris software package coupled with standard cell biology, biochemistry and molecular biology techniques in the lab.
This group have shown that lipofuscin bisretinoids trap cholesterol in RPE late endosomes and lysosomes. More recently, they reported that lipofuscin bisretinoids and cholesterol interfere with autophagosome trafficking and autophagic flux by activating acid sphingomyelinase (ASMase) in RPE lysosomes.
The Lakkaraju lab is one of many labs that chose the Revolution XD for this type of application because "Organelle movements in living cells are rapid and three dimensional, requiring continuous image capture through the Z plane for downstream time-based analysis”. Their lab uses the Andor Revolution XD system with the micro-lens based spinning disk because it offers gentler illumination, resulting in negligible light damage to the sample even over long imaging intervals. In addition, raster scanning with multiple pinholes allows for high-speed imaging at low light levels. The emitted light is sent to an EMCCD camera, which has greater quantum efficiency than PMTs, and is essential for low-light imaging. The Revolution XD can take images through the cell volume at speeds of 30-50 frames/second, and image multiple populations of cells simultaneously. These features are critical for imaging fast intracellular organelle trafficking in living cells without damaging the specimens.

L to R: Li Xuan Tan, Aparna Lakkaraju and Kimberly Toops
Biography
Aparna Lakkaraju was born in Bombay (now Mumbai) and raised in Hyderabad, India. After earning a Bachelors degree in Pharmaceutical Sciences from Kakatiya University, she came to the United States for graduate school. She worked with Dr. Janet Dubinsky and Dr. Yueh-Erh Rahman at the University of Minnesota, Minneapolis, where she developed a novel liposome-based gene delivery system for neurons.
Studying how these liposomes traffic in neurons spurred her interest in cell biology and she decided to do a post-doctoral fellowship in cell biology and ophthalmology in the laboratory of Dr. Enrique Rodriguez-Boulan at Cornell University’s Weill Medical College in New York City. In New York, she began her foray into vision research and was struck by how little is known about the basic biology of complex diseases like age-related macular degeneration. This led her to focus her research program on the cell biology of the retinal pigment epithelium and investigate how functional deficits in intracellular trafficking and cholesterol homeostasis can contribute to chronic dysfunction and disease.
Recent Publications
